Superfluids and
superconductors
Superfluids Helium-4 is a very peculiar liquid because it behaves strangely at temperatures below 2.17 K.
Imagine a beaker of the liquid on a turntable. If the turntable is rotated at steady speed, the liquid in the beaker eventually attains the same speed of rotation ‑ provided its temperature is between its boiling point (which is 4 K) and 2.17 K. However, if its temperature is less than 2.17 K, the liquid does not even begin to rotate. Its viscosity has disappeared so the rotating beaker does not affect it.
• Even stranger, a film of the liquid will creep up the inside of the beaker and down the outside. The liquid pours itself out of the beaker!
• The fountain effect is even more spectacular. A flashlight causes a jet of liquid helium to shoot into the air from a narrow tube projecting out of the liquid.
Below 2.17 K, helium4 is called a superfluid because of its strange behaviour. The temperature at which the transition takes place, 2.17 K, is called the lambda point because the specific heat capacity v temperature curve of liquid helium near the transition temperature is shaped like the Greek letter l. The curve has a discontinuity at the lambda point.
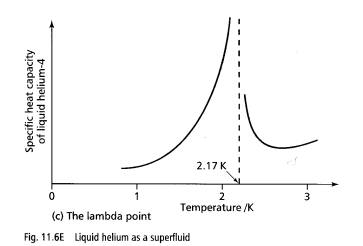
Why does hellum-4 below 2.17 K behave in this way? Scientists think that below the lambda point, the atoms are moving slowly enough to link together in a single energy state. Effects such as film creep, the fountain effect and frictionless flow arise because the atoms in a single energy state are unaffected by other atoms.
The lambda point represents a transition between disorder in which the atoms move about at random, and order in which the atoms move in a coordinated manner. Helium displays superfluidity because it remains in the liquid start at temperatures low enough for the atoms to form a single energy state.
Superconductors
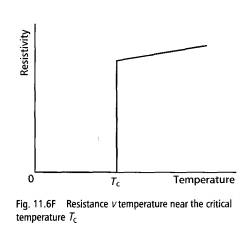
Superconducting effects A superconductor contains electrons that form a superfluid inside the superconductor. The property of superconductivity was discovered in 1911 when it was first observed that the electrical resistance of mercury disappears suddenly as the temperature is reduced below 4.15 K. Other elements as well as many alloys and compounds have been found to be superconducting at low temperatures. The temperature at which the resistance falls to zero is called the critical temperature Tc.
Until 1986, the highest critical temperature was 23.3 K. This was obtained with a compound of niobium and germanium.
The diagram shows how the resistivity of a superconductor varies near the metal's critical temperature.
Another strange effect occurs if a small magnet is placed on a sample of the material above its critical temperature. If the temperature of the sample is then reduced below 1c, the magnet rises above the sample. This happens because the magnetic field of the magnet was expelled from the sample when it became superconducting. This exclusion of magnetic flux from a superconductor is called the Meissner effect.
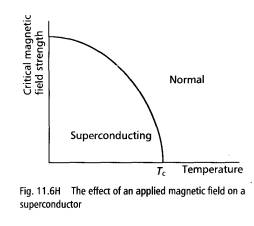
Superconductivity can be destroyed by an applied magnetic field. If a magnetic field is applied to a superconductor and increased from zero strength, the superconductivity is destroyed at certain field strength, called the critical field. Fig. 111.614 shows how the critical field strength varies with temperature.
High temperature superconductors In
1986, scientists discovered a class of materials which exhibited
superconductivity at 90 K, over 60 K higher than the existing record of 23.3 K.
More significantly, the critical temperature at 90 K is above the boiling point
of liquid nitrogen which is far cheaper than liquid helium. Since then, many
teams of scientists in different countries have been trying to discover
substances with higher critical temperatures and the current limit is now above
12 5 K. Any superconductor with a critical temperature above 77 K, the boiling
point of liquid nitrogen, is referred to as a high temperature superconductor.
Applications
of superconductors
An electromagnet with coils made of superconducting wires can create a very strong magnetic field because superconducting wires have zero resistance. This enables a very large current to pass through the wires without any resistance heating.
Superconducting power cables could transfer electrical power over vast distances without any heating effect in the cables. At present, up to 10% of the power transferred through a transmission cable is lost through the heating effect in the cable. However, too much current in a superconductor causes it to lose its superconductivity because of magnetic effects.
The current density in a current-carrying wire is defined as the current per unit cross-sectional area.
i.e. current density = current
cross-sectional area of the wire
At present, superconducting wires are capable of carrying current densities of up to 100 A/cm2.
Frictionless bearings are possible because of magnetic levitation. Magnetic levitation occurs when a superconductor is lowered onto a magnet and the superconductor floats above the magnet. The magnet induces a current in the superconductor which generates a magnetic field that opposes the magnet's field. The current doesn't decrease when the superconductor remains above the magnet because the superconductor has zero resistance so the current continues indefinitely.
Squids, or superconducting quantum interference devices, are used to detect very weak magnetic fields. A squid can even detect the magnetic fields produced by the brain! Fig. 11.6j shows the construction of a squid.
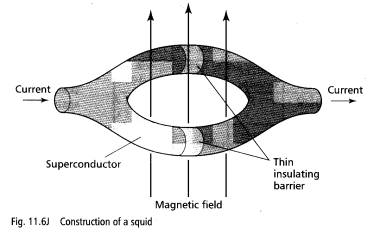
The insulating barrier in each branch is thin enough to allow pairs of electrons called Cooper pairs to 'tunnel' through. The current through the device can be changed by applying a weak magnetic field through the loop, as shown.
Cooper pairs Superconductivity in metals at very low temperatures is thought to arise as a result of conduction electrons separated by 100 atoms or more pairing together. Such pairs, known as Cooper pairs, are like the atoms in superconducting helium in that they are in a single energy state. They collide elastically with lattice atoms because the lattice atoms are unable to supply sufficient energy to break a pair. Hence Cooper pairs move through the metal without energy loss.
High temperature superconductors are complicated compounds, usually in ceramic form, and are not yet understood. Experimental physicists in laboratories in every continent are using 'trial and error' methods to improve superconductors. Theoretical physicists are striving to develop a theory for high temperature superconductors. The story of superconductivity illustrates the revolutionary and exciting nature of physics as a subject with immense benefits!